We wish to congratulate Dr. Miriam Heibeler -
with Head of department Prof. Dr. Oliver Reich at Klinikums
Harlaching Lehrkrankenhaus der Ludwig-Maximilians-Universität München for
her comprehensive thesis work on Synergo® published in 2020. We are extremely proud to share data from her study (translated from German). Full text: https://edoc.ub.uni-muenchen.de/25860/1/Hiebeler_Miriam.pdf
The following is a summary translated from Dr. Hiebeler study and with her permission.
The intended information is for professionals only.
Microwave-Induced Hyperthermia in combination
with Chemotherapy:
A retrospective analysis from Harlaching Hospital (2009-2015)
Munich, Germany
Dr. Miriam Hiebeler
A total of 44 non-muscle invasive bladder cancer patients were treated with Synergo® in München Harlaching Hospital Munich between 2009-2015. 41% of the patients were treated according to the prophylactic (adjuvant) protocol and 59% treated according to Synergo® ablative (neo-adjuvant) protocol.
Synergo prophylactic (adjuvant):
18/44 (41%) intermediate and high-risk patients, of which 50% had history of recurring disease with an average of 2.3 incidences prior to Synergo therapy.
Results: At the end of Synergo® treatment cycle, all patients but 2 (11%), who were lost to follow-up, were tumor-free.
14 of the 16 patients were tumor-free after completion of the Synergo therapy. This corresponds to 87.5% of the patients. The 2 patients who had a recurrence (mean time 2.3 years) both had pTaG1 tumor prior to Synergo which presented itself also when recurrence was recorded.
The follow-up period for this cohort lasted an average of 3.2 years.
No cystectomy had to be performed during that period.


Synergo ablative (neo-adjuvant):
26/44 (59%) of the patients were treated. 90% were classified as high-risk (according to the EAU guidelines), with TaG3, T1G2, T1G3 and/or CIS. 73% of the patients thad history of recurring disease.
An analysis of two groups who received Synergo ablative was performed. One group (62%) treated according to the protocol and the other (38%) was given Synergo as a therapy attempt although patients had contraindications prior to treatment (e.g. history of other tumours or urothelial carcinoma of the upper urinary tract).
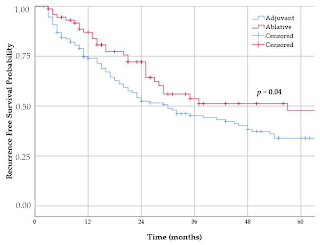
The Kaplan-Meier curve of all patients treated according to the ablative protocol shows 83% disease-free at one year and 70% at disease-free 5 years.

Below are the two ablative cohorts analysed separately:
Results of ablative Synergo:
16 patients were treated according to the Synergo recommended protocol. 15/16 of the patients were classified high-risk (according to EAU), one patient was classified as intermediate-risk. 73% of the patients had a history of disease and several were highly recurrent (3 or more episodes in the previous two years). 6 patients were “first episode” who had been diagnosed with T1 or high-grade carcinoma. Five of the latter had concomitant carcinoma in situ (CIS).
Of the 16 patients treated, 15 (93.8%) patients showed a complete response rate (tumor-free urinary bladder biopsies at the end of the ablation series). The average time to complete response was 1.9 months.
In one patient, the ablation therapy had no effect and a progression from TaG3 to pT2aG2 was recorded. This patient was cystectomised. Patients with complete response continued treatment and follow-up. The mean follow-up time was 24.2 months.
13/15 (86.7%) of the patients remained disease free for an average of 26 months and showed no further tumor formation during this period. Two patients (13.3%) had tumor recurrence. They were ultimately cystectomised after a follow-up period of 24.2 months.
In total, four of the 16 patients had to have their bladder removed. Two patients suffered tumor recurrence, one patient showed tumor progression and a cystectomy was performed on another patient with a reduced bladder capacity of less than 90 ml. The resected bladder, however, was completely tumor-free. One patient was partially resected because of a suspected tumor recurrence. The resected material was tumor-free. In the subsequent examinations, no new tumor was detected in the rest of the urinary bladder.

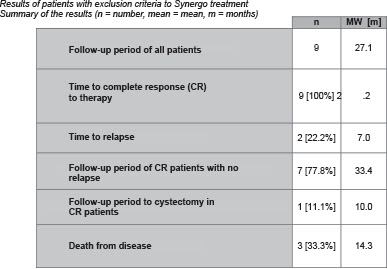
Results of ablative Synergo in patients with known exclusion criteria for Synergo 10/26):
One patient stopped therapy prematurely due to severe allergic skin reactions. His therapy results were not taken into account.
7/9 (77.8%) showed no tumor recurrence or progression after a follow-up period of an average of 33.4 months.
Two patients (22.2%) experienced recurrence in the form of muscle-invasive carcinoma. One developed pT2G3 carcinoma from TaG3 stage, the other developed invasive pT3aG3 carcinoma from T1G3 tumor. This patient (11.1%) underwent a cystectomy ten months after start of Synergo therapy.
A total of three patients (33.3%) died as a result of bladder carcinoma. In two patients the urinary bladder was tumor-free at the time of death; they died from extensive metastasis. The average time to death was 14.3 Months.
Side effects to therapy:
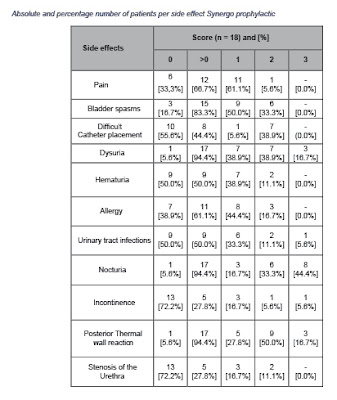
Side effects noted in a total of 63% of the prophylactic treated patients and in 69% of the ablative treated patients.
The most common were pain in the form of bladder cramps during treatments, dysuria, nocturia and allergy. Severe complications in the form of urethral strictures occurred in 27.8% of the patients, although there was no stricture impeding a cystoscope to pass. The most noted side effect observed in almost all patients during cystoscopy was posterior thermal wall reaction (average grade 2 in both groups). The posterior thermal wall reaction is symptom-free and disappears after a few months. Below are the detailed tables of side effects recorded in both prophylactic and ablative Synergo treatments.

Medical Disclaimer
While clinical studies support the safety and effectiveness of the Synergo® RF thermo-chemotherapy system when used in the treatment of non-muscle invasive bladder cancer, results may vary. There are no guarantees of outcome. Before you decide on treatment options, discuss them with your doctor. Understanding the risks of each treatment can help you make the best decision for your individual situation. Synergo treatment may not be appropriate for every individual; it may not be applicable to your condition. Always ask your doctor about all treatment options, as well as their risks and benefits. Only your doctor can determine whether Synergo is appropriate for your situation.